Q for folks who read a lot of #LongCovid studies-- do you know of any meta-analysis about the very diverse rates of long covid that are reported (1-40%)?
-
@beandreams If it exists, someone on S4ME will be able to point you to it. www.s4me.info
@eleanorrees Good call! That will be my next stop
-
@BE @longcovid Oh for sure, what I am curious about is whether there is a paper that tallies up the impact of all these decisions about definitions and study design
@beandreams @BE @longcovid just saw a post by @wecrunchme on instagram that seems to adress this topic. Quick printscreen.
I know him to answer questions
 Maybe you'll find more
Maybe you'll find more
https://crunchme.org/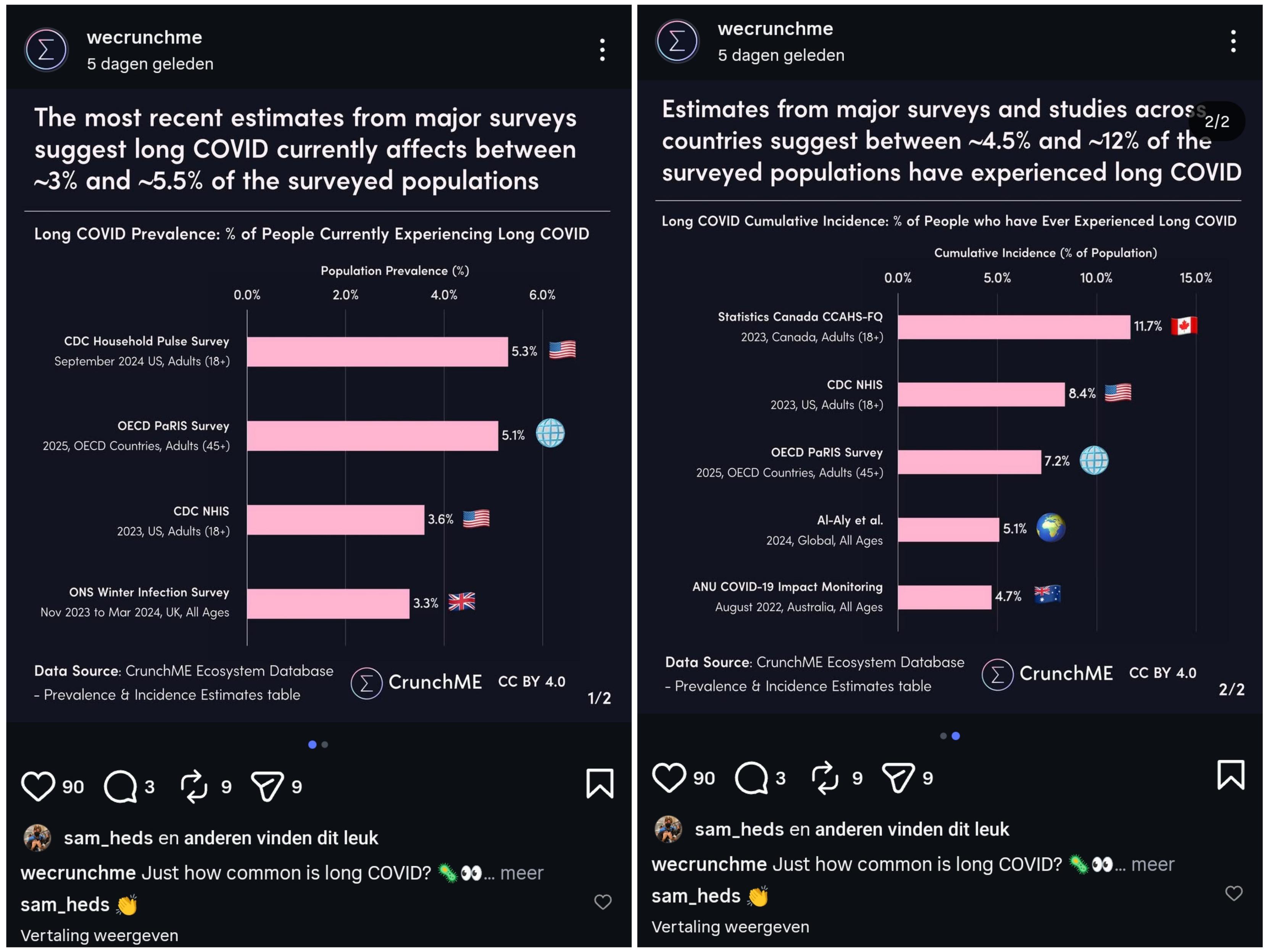
-
Without specific papers to look at, I'm guessing a little here, but it's probably down to how each paper defines long COVID.
You'll find pretty wildly different definitions across papers(at least as recently as a year ago when I read a lot more scientific papers on COVID).
@BE @beandreams @longcovid This is my take, too. Some will define long COVID as any new symptoms that fit the profile of long COVID (which probably leads to an overestimate of symptomatic prevalence), while others will only count actual diagnoses (a dramatic underestimate, IMO).
The other issue is that we're really only about 3 years into mass infection, which means we still have no idea how prevalent the long-term consequences of infection will be, nor how severe they could be. Much like how HIV can take a decade to become AIDS in some untreated patients.
My position is that everyone who has been infected has long COVID. It's just a matter of how fast their disease progresses and how symptomatic the damage is. There's also the issues that are not classified as long COVID (brain and cardiac damage, for example) that compound with every infection and have other knock-on systemic effects (people with cardiac disease lose 70% of T-cell function that doesn't recover after a full year vs. those without cardiac disease, whose T-cell function decline is 10% after a full year, for example).
-
@BE @beandreams @longcovid This is my take, too. Some will define long COVID as any new symptoms that fit the profile of long COVID (which probably leads to an overestimate of symptomatic prevalence), while others will only count actual diagnoses (a dramatic underestimate, IMO).
The other issue is that we're really only about 3 years into mass infection, which means we still have no idea how prevalent the long-term consequences of infection will be, nor how severe they could be. Much like how HIV can take a decade to become AIDS in some untreated patients.
My position is that everyone who has been infected has long COVID. It's just a matter of how fast their disease progresses and how symptomatic the damage is. There's also the issues that are not classified as long COVID (brain and cardiac damage, for example) that compound with every infection and have other knock-on systemic effects (people with cardiac disease lose 70% of T-cell function that doesn't recover after a full year vs. those without cardiac disease, whose T-cell function decline is 10% after a full year, for example).
@BE @beandreams @longcovid ALSO, self-reported "recovery" studies are exceptionally dubious, as people tend to psychologically adapt to new circumstances. Someone may experience no objective improvement in their health, but they'll often self-report improvement because they've learned to adapt and have forgotten what it was like to have good health.
-
@beandreams @BE @longcovid just saw a post by @wecrunchme on instagram that seems to adress this topic. Quick printscreen.
I know him to answer questions
 Maybe you'll find more
Maybe you'll find more
https://crunchme.org/
-
@BE @longcovid Oh for sure, what I am curious about is whether there is a paper that tallies up the impact of all these decisions about definitions and study design
@beandreams If you find such a paper, would you please @ me when you share it?
From bookmarks it seems I have:
Applying 5 published definitions for long COVID yielded a prevalence that ranged from 30.84% (95% CI, 29.33%-32.40%) to 42.01% (95% CI, 40.37%-43.66%) at 3 months and 14.23% (95% CI, 13.01%-15.55%) to 21.94% (95% CI, 20.47%-23.47%) at 6 months postinfection; in the 5 comparator studies, reported prevalence of long COVID at 1 to 5 months postinfection ranged from 2.6% (≥84 days) to 47.4% (3-5 months) and at 6 or more months postinfection ranged from 10.0% (95% CI, 8.8%-11.0%) to 61.9% (6-11 months). Using participants’ self-reported long COVID as a criterion standard, existing published definitions had low-to-moderate sensitivity (up to 66.32% [95% CI, 62.59%-69.90%] at 3 months and 45.53% [95% CI, 41.51%-49.60%] at 6 months) and high specificity (up to 81.29% [95% CI, 79.32%-83.15%] at 3 months and 94.26% [95% CI, 92.98%-95.37%]) at 6 months. [1]
Another recent-ish paper talked about the different trajectories Long COVID can take, with an unexamined implication being that studies might miss some trajectories due to sample timing and study length. Their work specifically avoided that:
A notable design strength of the RECOVER study is the inclusion of frequent serial measurements over time since initial infection from a population-based cohort. This design allowed us to apply finite mixture models for longitudinal data, an unbiased approach to characterizing distinct longitudinal profiles with additional robustness properties against overfitting. The observed eight longitudinal trajectories were heterogeneous across profiles, which is consistent with the reported clinical patient experience. [2]
A "mega" study unfortunately didn't look into the why very much, but stated
Studies varied substantially (I2 = 100%, P < .001), possibly due to heterogeneity in definition of long COVID, study designs and populations, evolution of SARS-CoV-2 and its variants from Alpha to Omicron subvariants, and testing and prevention/treatment strategies over a wide time span from 2021 to 2024. [3]
notably the range of Long COVID prevalence they found is extraordinary:
ranging from 3% to 80% for publications in 2024 [3]
and they cite suggestive statistical differences by location, symptom cluster/subtype, date, and so on, without really getting to the point that you (and I!) are interested in, which is why the studies have different rates.
[1] https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2837446
[2] https://www.nature.com/articles/s41467-025-65239-4
[3] https://pmc.ncbi.nlm.nih.gov/articles/PMC12461872/ -
@BE @beandreams @longcovid This is my take, too. Some will define long COVID as any new symptoms that fit the profile of long COVID (which probably leads to an overestimate of symptomatic prevalence), while others will only count actual diagnoses (a dramatic underestimate, IMO).
The other issue is that we're really only about 3 years into mass infection, which means we still have no idea how prevalent the long-term consequences of infection will be, nor how severe they could be. Much like how HIV can take a decade to become AIDS in some untreated patients.
My position is that everyone who has been infected has long COVID. It's just a matter of how fast their disease progresses and how symptomatic the damage is. There's also the issues that are not classified as long COVID (brain and cardiac damage, for example) that compound with every infection and have other knock-on systemic effects (people with cardiac disease lose 70% of T-cell function that doesn't recover after a full year vs. those without cardiac disease, whose T-cell function decline is 10% after a full year, for example).
@PacificNic just to add: T-cell decline is sometimes measured in undifferentiated T-cell counts, and because of feedback loops AJ Leonardi was suggesting that a 10% population drop in some T-cell types could cause significant dysregulation, which informs my mental model at least.
-
@PacificNic just to add: T-cell decline is sometimes measured in undifferentiated T-cell counts, and because of feedback loops AJ Leonardi was suggesting that a 10% population drop in some T-cell types could cause significant dysregulation, which informs my mental model at least.
@datum and it's borne out in the dramatic rise of autoimmune diseases in the last few years. Leonardi knew what he was talking about.
-
@beandreams If you find such a paper, would you please @ me when you share it?
From bookmarks it seems I have:
Applying 5 published definitions for long COVID yielded a prevalence that ranged from 30.84% (95% CI, 29.33%-32.40%) to 42.01% (95% CI, 40.37%-43.66%) at 3 months and 14.23% (95% CI, 13.01%-15.55%) to 21.94% (95% CI, 20.47%-23.47%) at 6 months postinfection; in the 5 comparator studies, reported prevalence of long COVID at 1 to 5 months postinfection ranged from 2.6% (≥84 days) to 47.4% (3-5 months) and at 6 or more months postinfection ranged from 10.0% (95% CI, 8.8%-11.0%) to 61.9% (6-11 months). Using participants’ self-reported long COVID as a criterion standard, existing published definitions had low-to-moderate sensitivity (up to 66.32% [95% CI, 62.59%-69.90%] at 3 months and 45.53% [95% CI, 41.51%-49.60%] at 6 months) and high specificity (up to 81.29% [95% CI, 79.32%-83.15%] at 3 months and 94.26% [95% CI, 92.98%-95.37%]) at 6 months. [1]
Another recent-ish paper talked about the different trajectories Long COVID can take, with an unexamined implication being that studies might miss some trajectories due to sample timing and study length. Their work specifically avoided that:
A notable design strength of the RECOVER study is the inclusion of frequent serial measurements over time since initial infection from a population-based cohort. This design allowed us to apply finite mixture models for longitudinal data, an unbiased approach to characterizing distinct longitudinal profiles with additional robustness properties against overfitting. The observed eight longitudinal trajectories were heterogeneous across profiles, which is consistent with the reported clinical patient experience. [2]
A "mega" study unfortunately didn't look into the why very much, but stated
Studies varied substantially (I2 = 100%, P < .001), possibly due to heterogeneity in definition of long COVID, study designs and populations, evolution of SARS-CoV-2 and its variants from Alpha to Omicron subvariants, and testing and prevention/treatment strategies over a wide time span from 2021 to 2024. [3]
notably the range of Long COVID prevalence they found is extraordinary:
ranging from 3% to 80% for publications in 2024 [3]
and they cite suggestive statistical differences by location, symptom cluster/subtype, date, and so on, without really getting to the point that you (and I!) are interested in, which is why the studies have different rates.
[1] https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2837446
[2] https://www.nature.com/articles/s41467-025-65239-4
[3] https://pmc.ncbi.nlm.nih.gov/articles/PMC12461872/@datum Thank you for these! I really appreciate folks like you who are persevering to keep an eye on the new work coming out and can give highlights! These days I really only glance at new papers to check whether they mean I need to update my mental model or not.
And yeah, that range! If you included some of the brain scan or cognitive decline studies for the high end, and some of the badly designed diagnosis-only ones for the low, I bet you could find a reported range of 1-99% lol
-
@datum Thank you for these! I really appreciate folks like you who are persevering to keep an eye on the new work coming out and can give highlights! These days I really only glance at new papers to check whether they mean I need to update my mental model or not.
And yeah, that range! If you included some of the brain scan or cognitive decline studies for the high end, and some of the badly designed diagnosis-only ones for the low, I bet you could find a reported range of 1-99% lol
@datum There's this funny side-effect of that range too, which is that it makes rates like 2% seem low. If there was an adverse side-effect of a drug that occurred 2% of the time, it would be labelled "common"
-
R relay@relay.mycrowd.ca shared this topic
